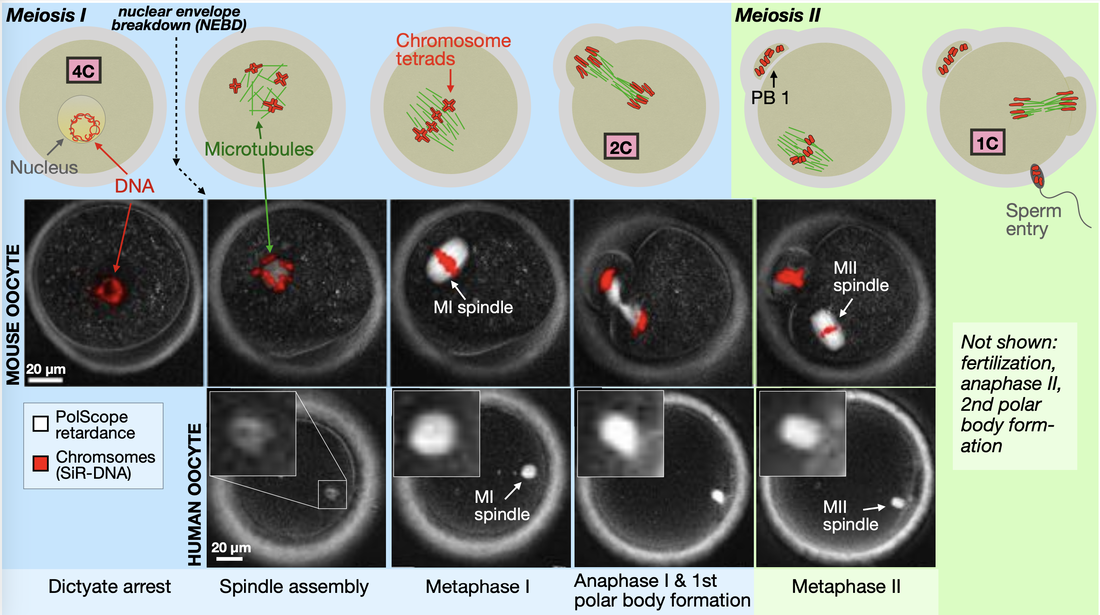
Imaging spindle structure in living mouse and human oocytes. Oogenesis in mammals comprises two sequential cell divisions, meiosis I and meiosis II, each of which produces a relatively large, steady-state spindle that persists for several hours. In the mouse oocyte, DNA is labeled with a fluorescent dye; in the human oocyte, DNA is unlabeled. In both samples, spindles are imaged with PolScope. At nuclear envelope breakdown (NEBD), the oocyte contains four copies of each chromosome (4C), which condense and form chromosome tetrads. During each meiotic division, the amount of DNA in the oocyte is reduced by half. In mouse, the MII spindle forms ~10 hrs after NEBD. In human, the same process takes ~20 hrs. In both species, fully formed MI and MII spindles persist for many hours.
Confocal Image of a Living Oocyte Spindle The confocal image on the right shows three of the important structural subunits of the spindle: microtubules, chromosomes, and kinetochores. The spindle contains several categories of active and passive cross-linkers (molecules that create forces between microtubules); these are not imaged. Microtubules are labeled with a small-molecule dye; kinetochores and chromosomes are labeled with a genetically engineered fluorescent protein, introduced to the oocyte via mRNA microinjection.
|
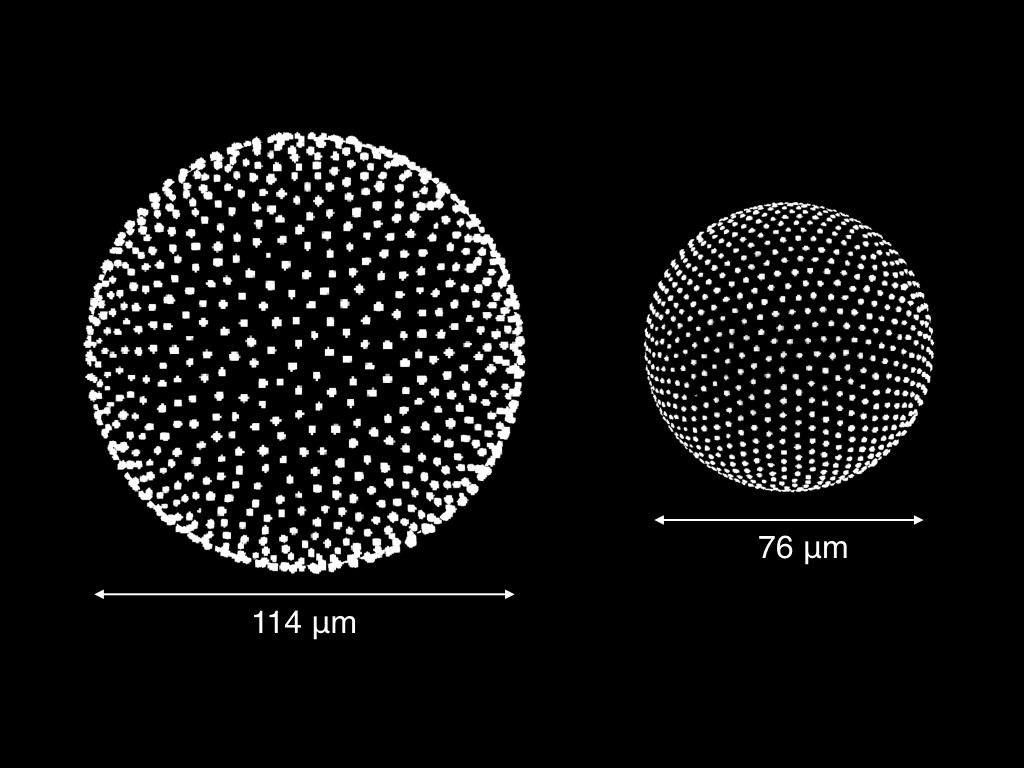
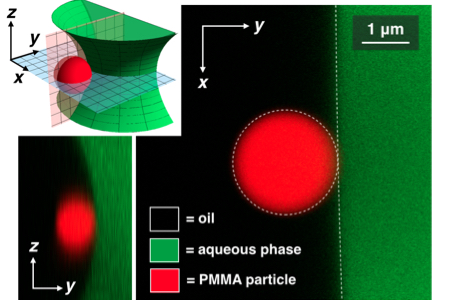
Charged particles on spherical droplets (3D rendering of confocal micrograph) In the image on the left, the white dots are fluorescent PMMA particles, which bind to the interior surface of a spherical oil droplet. At low density, the system appears disordered (left droplet). However, as the spacing between the particles is reduced, the system becomes more ordered (right droplet). How can we describe this apparent phase behavior?
|
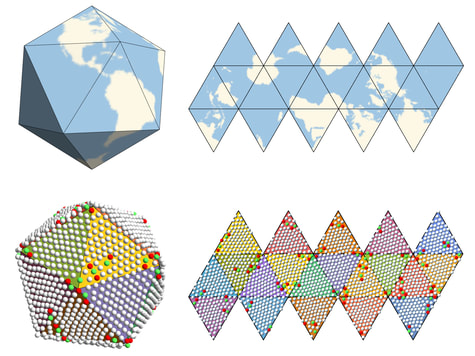
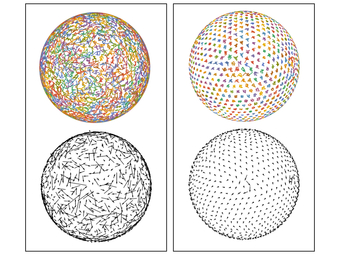
Unfolding an icosahedral projection. By using an icosahedral projection to map the particle positions to flat space, the long-range orientational order in the system becomes apparent.
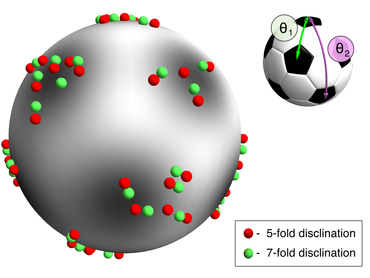
Fitting the defect positions. By using an appropriate combination of spherical harmonics, we can find the best global fit of the defect particles to the vertices of an icosahedron.