PhD work
My PhD research centered on using colloidal particles to understand basic questions in condensed matter physics. Colloids are made up of microscopic particles which are dispersed in fluid, for instance water or oil. Many common household products - for example milk, coffee, and sunscreen - are colloidal in nature. From the perspective of a physicist, one of the cool things about colloids is that they are both small enough to reach thermal equilibrium in reasonable times (unlike baseballs), and are large enough that they can be seen with an optical microscope (unlike atoms). This makes colloids ideal as a model system for studying basic questions about how matter organizes itself under various conditions.
During my PhD, I studied questions like: how do two dimensional materials melt? What is the role of geometry in determining order on the micro-scale? How does background curvature affect phase behavior? What are the similarities and differences between the colloidal glass transition in 2D and 3D?
During my PhD, I studied questions like: how do two dimensional materials melt? What is the role of geometry in determining order on the micro-scale? How does background curvature affect phase behavior? What are the similarities and differences between the colloidal glass transition in 2D and 3D?
|
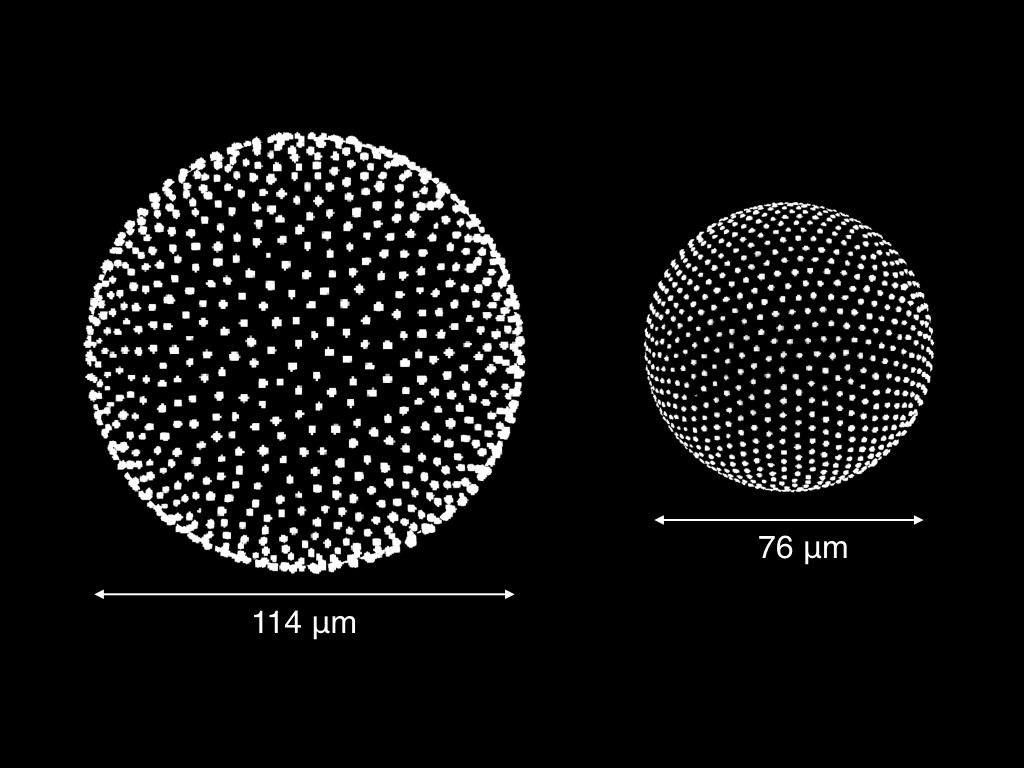
Charged particles on spherical droplets (3D rendering of confocal micrograph) In the image on the left, the white dots are fluorescent PMMA particles, which bind to the interior surface of a spherical oil droplet. At low density, the system appears disordered (left droplet). However, as the spacing between the particles is reduced, the system becomes more ordered (right droplet). How can we describe this apparent phase behavior?
|